Encrypted code: Hyperexcitability of the trigeminal system Abstract
Encrypted code: Hyperexcitability of the trigeminal system
Abstract
This chapter introduces the Cognitive Neural Network (CNN) as a practical clinical instrument to navigate diagnostic uncertainty in complex orofacial disorders, using the patient known as “Bruxer” as an exemplar. Bruxer presents with severe nocturnal and diurnal bruxism, chronic orofacial pain (OP), and a set of neurological signs that cannot be safely explained by a purely dental model. The aim of this section is to demonstrate how the CNN—already presented in the chapter “Encrypted Code: Ephaptic Transmission”—can be re-applied as a repeatable procedure to “decrypt” the machine-language output of the Central Nervous System (CNS) and translate it into a coherent diagnostic direction.
The point of departure is the preceding phase of the Masticationpedia method: the discrimination of contexts via the coherence demarcator . In the Bruxer case, the dental context is largely negative (radiology, axiography, baseline EMG symmetry), while the neurological context is weakly positive, driven mainly by a modest amplitude asymmetry in the jaw jerk. This produces a borderline diagnostic slope () that is insufficient, by itself, to justify decisive clinical action. The CNN is therefore used to refine the search space: it operationalizes the clinician’s reasoning by structuring a sequence of increasingly selective PubMed queries, progressing from broad terms (bruxism) to targeted neurophysiological descriptors (trigeminal system, abnormality, excitability). This guided narrowing is not a bibliographic exercise, but a cognitive filter designed to identify the smallest set of electrophysiological mechanisms compatible with the patient’s observed “code.”
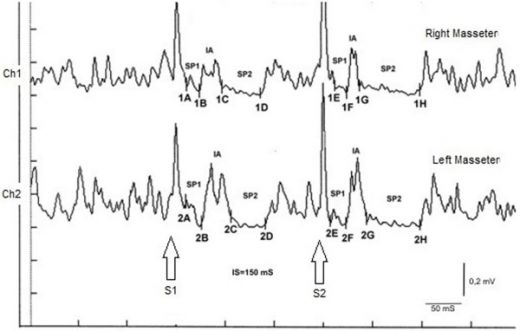
The CNN sequence converges on a specific hypothesis: abnormal excitability of trigeminal motor pathways in sleep bruxism, likely mediated by subcortical and brainstem inhibitory circuits rather than purely cortical mechanisms. On this basis, the chapter selects a decisive functional test: the recovery cycle of the Masseter Inhibitory Reflex (rcMIR). The rcMIR protocol is described as a paired-stimulus paradigm (conditioning S1 and test S2) designed to evaluate how quickly inhibitory brainstem circuitry recovers and whether a second stimulus can evoke a normal or pathological silent period pattern. In Bruxer, the S2 stimulus evokes both silent periods with abnormal persistence and altered interposed activity, a pattern that is inconsistent with normal inhibitory recovery and strongly indicative of trigeminal system hyperexcitability.
This “decrypted” signal—hyperexcitability—becomes the pivot of the diagnostic pathway. It triggers an urgent escalation from functional testing to structural imaging. A brain MRI then reveals a pineal cavernoma with associated signs of ventricular involvement and transependymal fluid absorption, providing a definitive organic correlate for the patient’s electrophysiological abnormalities. The chapter concludes that severe bruxism, particularly when coupled with OP and subtle neurological signs, must be treated as a potential manifestation of CNS dysfunction and not as a purely dental parafunction. In this framework, “bruxism” remains a vague verbal label, while “hyperexcitability” represents a higher-fidelity translation of CNS machine language—actionable, measurable, and diagnostically orienting. The CNN therefore functions as a repeatable clinical compass: it reduces ambiguity, improves safety in differential diagnosis, and supports earlier identification of organic disease hidden beneath apparently dental symptoms.
🧠 Three guiding questions (with essential answers)
1️⃣ When should bruxism no longer be interpreted as a dental disorder but as a manifestation of central nervous system dysfunction? – When dental findings are negative or of low clinical severity while neurological signs emerge, such as generalized hyperreflexia, visual disturbances, nystagmus, abnormal jaw jerk amplitude, or widespread muscular stiffness. In this context, bruxism becomes a verbal label masking an underlying condition of central hyperexcitability.
2️⃣ Which neurophysiological finding allows the decryption of the vague term “bruxism” into a measurable clinical entity? – The demonstration of abnormal excitability within the trigeminal motor system, particularly through the Recovery Cycle of the Masseter Inhibitory Reflex (rcMIR). An abnormal recovery pattern indicates instability of inhibitory brainstem circuits and provides objective evidence of central hyperexcitability.
3️⃣ What diagnostic step definitively closes the loop between functional symptoms and organic pathology in complex bruxism cases? – Targeted brain MRI following neurophysiological evidence of central hyperexcitability. In this case, imaging revealed a pineal cavernoma, transforming an apparently functional or dental problem into a clearly defined neurological diagnosis and preventing further diagnostic delay.
If you already have an approved account, click on your profile icon and return to the Book Index to read the complete chapter.
To access the full content, you must sign in via LinkedIn and request approval.
Bibliography & references
Peter H Yang; Alison Almgren-Bell; Hongjie Gu; Anna V Dowling; Sangami Pugazenthi; Kimberly Mackey; Esther B Dupépé; Jennifer M Strahle, «Etiology- and region-specific characteristics of transependymal cerebrospinal fluid flow», 2022».
Frisardi G, «The use of transcranial stimulation in the fabrication of an occlusal splint», 1992».
Frisardi G; Ravazzani P; Tognola G; Grandori F, «Electric versus magnetic transcranial stimulation of the trigeminal system in healthy subjects. Clinical applications in gnathology», 1997».
von Essen C; Rydenhag B; Nystrom B; Mozzi R; van Gelder N; Hamberger A, «High levels of glycine and serine as a cause of the seizure symptoms of cavernous angiomas?», 1996».
Lau A; Tymianski M, «Glutamate receptors, neurotoxicity and neurodegeneration», 2010».
Meldrum B; Garthwaite J, «Excitatory amino acid neurotoxicity and neurodegenerative disease», 1990».
Xiao Y; Richter JA; Hurley JH, «Release of glutamate and CGRP from trigeminal ganglion neurons: role of calcium channels and 5-HT1 receptor signaling», 2008».
McCleskey EW; Fox AP; Feldman DH; Cruz LJ; Olivera BM; Tsien RW; Yoshikami D, «Omega-conotoxin: direct and persistent blockade of specific types of calcium channels in neurons but not muscle», 1987».
Hockerman GH; Johnson BD; Abbott MR; Scheuer T; Catterall WA, «Molecular determinants of high affinity phenylalkylamine block of L-type calcium channels in transmembrane segment IIIS6 and the pore region of the alpha1 subunit», 1997».
Tallaksen-Greene SJ; Young AB; Penney JB; Beitz AJ, «Excitatory amino acid binding sites in the trigeminal principal sensory and spinal trigeminal nuclei of the rat», 1992».
Storer RJ; Goadsby PJ, «Trigeminovascular nociceptive transmission involves N-methyl-D-aspartate and non-N-methyl-D-aspartate glutamate receptors», 1999».
Goadsby PJ; Classey JD, «Glutamatergic transmission in the trigeminal nucleus assessed with local blood flow», 2000».
Waeber C; Moskowitz MA; Cutrer FM; Sanchez Del Rio M; Mitsikostas DD, «The NMDA receptor antagonist MK-801 reduces capsaicin-induced c-fos expression within rat trigeminal nucleus caudalis», 1998».
Knight YE; Bartsch T; Kaube H; Goadsby PJ, «P/Q-type calcium-channel blockade in the periaqueductal gray facilitates trigeminal nociception: a functional genetic link for migraine?», 2002».
Dunlap K; Luebke JI; Turner TJ, «Exocytotic Ca2+ channels in mammalian central neurons», 1995».
Pan ZZ; Williams JT; Osborne PB, «Opioid actions on single nucleus raphe magnus neurons from rat and guinea-pig in vitro», 1990».
Hentall ID, «Interactions between brainstem and trigeminal neurons detected by cross-spectral analysis», 2000».
Jiang M; Behbehani MM, «Physiological characteristics of the projection pathway from the medial preoptic to the nucleus raphe magnus of the rat and its modulation by the periaqueductal gray», 2001».
Cruccu G; Berardelli A; Inghilleri M; Manfredi M, «Functional organization of the trigeminal motor system in man. A neurophysiological study», 1989».
Li YQ; Takada M; Kaneko T; Mizuno N, «GABAergic and glycinergic neurons projecting to the trigeminal motor nucleus: a double labeling study in the rat», 1996».
Bourque MJ; Kolta A, «Properties and interconnections of trigeminal interneurons of the lateral pontine reticular formation in the rat», 2001».
McDavid S; Verdier D; Lund JP; Kolta A, «Electrical properties of interneurons found within the trigeminal motor nucleus», 2008».
Kolta A; Westberg KG; Lund JP, «Identification of brainstem interneurons projecting to the trigeminal motor nucleus and adjacent structures in the rabbit», 2000».
Ongerboer de Visser BW; Cruccu G; Manfredi M; Koelman JH, «Effects of brainstem lesions on the masseter inhibitory reflex. Functional mechanisms of reflex pathways», 1990».
Svensson P; Jadidi F; Arima T; Baad-Hansen L; Sessle BJ, «Relationships between craniofacial pain and bruxism», 2008».
Watts MW; Tan EK; Jankovic J, «Bruxism and cranial-cervical dystonia: is there a relationship?», 1999».
Monaco A; Ciammella NM; Marci MC; Pirro R; Giannoni M, «The anxiety in bruxer child. A case–control study», 2002».
Lobbezoo F; Visscher CM; Naeije M, «Some remarks on the RDC/TMD Validation Project: report of an IADR/Toronto-2008 workshop discussion», 2010».
Frisardi G; Chessa G; Sau G; Frisardi F, «Trigeminal electrophysiology: a 2×2 matrix model for differential diagnosis between temporomandibular disorders and orofacial pain», 2010».
