1° Clinical case: Hemimasticatory spasm
Abstract
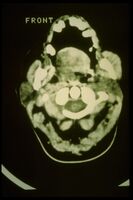
This chapter continues the diagnostic series by using the case of “Mary Poppins” to show why complex orofacial disorders cannot be solved inside a single disciplinary frame. After a decade of uncertainty—multiple dental investigations, neurological consultations, imaging studies, and electrophysiological tests—the patient’s clinical picture demonstrates a recurring problem in real-world medicine: two different specialist contexts may each appear internally coherent, yet lead to opposite diagnostic conclusions. In Mary Poppins, standard dental data (temporomandibular joint remodeling, occlusal alterations, asymmetries on instrumental exams) can support a temporomandibular disorder narrative, while neurophysiological abnormalities (jaw jerk and masseteric silent period alterations, conduction delays, neuromotor signs) support a neuromotor disorder narrative. The chapter formalizes this conflict as a “context collision” between a dental proposition set and a neurological proposition set, each consistent with its own reported findings.
To resolve this diagnostic deadlock, the chapter introduces a demarcation strategy: a clinical “coherence marker” that assigns different diagnostic weight to positive findings according to their severity and pathophysiological specificity. In practice, not all “positive” tests carry equal inferential power. Structural joint remodeling may be clinically relevant but often nonspecific, while a clear electrophysiological absence or latency shift in a brainstem-related trigeminal reflex can represent a stronger indicator of neurological involvement. By applying as a severity-weighting operator, the clinician can filter out low-specificity interference and identify the dominant diagnostic trajectory. This formal step does not replace clinical reasoning; it makes reasoning explicit, transparent, and reproducible when multidisciplinary statements overlap.
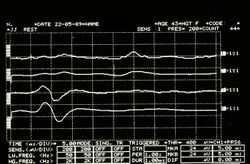
Once the demarcation establishes a neuromotor priority, the chapter moves from “what is consistent” to “what mechanism can explain it,” focusing on peripheral nerve conduction and the possibility of focal demyelination. The diagnostic pathway therefore advances to targeted trigeminal electrophysiology: stimulation of the masseter nerve in the infratemporal fossa to evoke the -wave, paired with bilaterally evoked trigeminal motor root responses (Root-MEPs). The documented conduction delay on the symptomatic side—without proportional loss of amplitude—raises suspicion for axon-sparing demyelination rather than primary myopathy. The investigation is then refined by searching for heteronymous responses (the -wave recorded on the temporalis muscle) to determine whether abnormal excitability spreads beyond the directly stimulated pathway.
At the core of the chapter lies the concept that becomes the “encrypted key” of the case: ephaptic transmission. Ephaptic mechanisms describe pathological electrical coupling between adjacent nerve fibers, typically facilitated by demyelination, where impulses can spread laterally and generate ectopic excitation and synchronized discharges. This mechanism offers a coherent explanation for the patient’s involuntary masticatory activity, the neurophysiological asymmetries, and the paradoxical coexistence of dental consequences with a primary neuropathic origin. The chapter closes by linking these findings to clinical management: botulinum toxin therapy directed at the hyperactive muscles, guided by neurophysiological localization, can achieve long-term symptom control. Beyond the single case, the chapter argues for a paradigm shift: diagnostics must tolerate graded uncertainty (fuzzy logic), integrate probabilistic thinking, and treat “borderline” syndromes as system-level phenomena rather than isolated dental or neurological labels.
🧠 Three guiding questions (with essential answers)
1️⃣ Why can Mary Poppins’ clinical picture generate two “coherent” but opposite diagnoses (TMD vs neuromotor disorder)? — Because the same patient can produce two internally consistent sets of propositions: dental findings () support a temporomandibular narrative (), while neurophysiological findings () support a neuromotor narrative (). Without a weighting rule, both unions () and () can appear “true,” causing diagnostic delay.
2️⃣ What is the function of the coherence marker in the chapter’s diagnostic logic? — assigns different diagnostic weight to “positive” findings according to severity and pathophysiological specificity, filtering low-specificity interference and preventing context collision. In practice it prioritizes high-impact neurophysiological abnormalities (e.g., absent/prolonged jaw jerk or silent period) over nonspecific structural TMJ changes, allowing the demarcation step () to select the dominant diagnostic pathway.
3️⃣ Why is ephaptic transmission the “encrypted key” that explains Hemimasticatory Spasm in this case? — Because focal demyelination can enable pathological lateral spread of impulses between adjacent trigeminal motor fibers, producing ectopic excitation, synchronized high-frequency discharges, and involuntary spasms without a normal synaptic pathway. This mechanism coherently links conduction delay with preserved amplitude (axon-sparing demyelination), abnormal reflex physiology, and the spread of activity from masseter pathways toward temporalis involvement.
If you already have an approved account, click on your profile icon and return to the Book Index to read the complete chapter.
To access the full content, you must sign in via LinkedIn and request approval.
Bibliography & references
- Jankovic J. Peripherally induced movement disorders. Neurol Clin. 2009;27(3):821–832. DOI: 10.1016/j.ncl.2009.04.005.
- Lee SH, Rhee BA, Choi SK, Koh JS, Lim YJ. Cerebellopontine angle tumors causing hemifacial spasm: types, incidence, and mechanism in nine reported cases and literature review. Acta Neurochir (Wien). 2010;152(11):1901–1908. DOI: 10.1007/s00701-010-0796-1.
- Peker S, Ozdamar K, Kilic T, Pamir MN. Relief of hemifacial spasm after radiosurgery for intracanalicular vestibular schwannoma. Minim Invasive Neurosurg. 2004;47(4):235–237. DOI: 10.1055/s-2004-818486.
- Bertot DB, Le Presti M, Stormes K, Raskin JS, Lea A, Chelius D, Lams D. Trigeminal schwannoma presenting with malocclusion: A case report and review of the literature. Surg Neurol Int. 2020;11:230. DOI: 10.25259/SNI_482_2019.
- Agarwal A. Intracranial trigeminal schwannoma. Neurology. 2015;84(21):36–41. DOI: 10.1212/WNL.0000000000002086.
- Kaminska J, Koper OM, Piecha K, Kernow H. Multiple sclerosis – etiology and diagnostic potential. Postępy Hig Med Dosw. 2017;71:551–563. DOI: 10.5604/01.3001.0010.3836.
- Yates SK, Brown WF. The human jaw jerk: electrophysiological changes in multiple sclerosis. Neurology. 1981;31(5):632–634. DOI: 10.1212/WNL.31.5.632.
- Koyuncu M, Sezen T, Akan H, Ismailoglu AA, Tanyeri Y, Teka A, Unal R. Incidental of computed tomography and magnetic resonance imaging in the diagnosis of parotid tumors. Oral Radiol. 2003;19(2):76–732. DOI: 10.1016/j.clinimag.2003.07.009.
- Machado RA, Moubayed SPM, Khorsandi A, Hernandez-Perez JC, Urken ML. Intermittent facial spasm as the presenting sign of a recurrent pleomorphic adenoma. World J Clin Oncol. 2017;8(1):86–90. DOI: 10.5306/wjo.81.86.
- Cruccu G, Truini A, Priori A. Excitability of the human trigeminal motoneuronal pool and interactions with other brainstem reflex pathways. J Physiol. 2001;531(Pt 2):559–571. DOI: 10.1111/j.1469-7793.2001.0559i.x. PMCID: PMC2278464.
- Frisardi G. The use of transcranial stimulation in the fabrication of an occlusal splint. J Prosthet Dent. 1992;68(2):345–350. DOI: 10.1016/0022-3913(92)90345-B.
- Frisardi G, Ravazzani P, Tognola G, Grandori F. Electric versus magnetic transcranial stimulation of the trigeminal system in healthy subjects. J Oral Rehabil. 1997;24(12):920–928. PMID: 9467995.
- Nielsen VK. Pathophysiology of hemifacial spasm: I. Ephaptic transmission and ectopic excitation. Neurology. 1984;34(4):418–426. PMID: 6322048.