Hemimasticatory spasm
Abstract
This chapter reconstructs the diagnostic reasoning that leads from a generic label of “masticatory dysfunction” to a precise neuromotor diagnosis: Hemimasticatory Spasm (HMS), presented through the clinical pathway of a patient referred to as “Mary Poppins.” Although the phenomenology of HMS—unilateral involuntary contractions of masticatory muscles—may appear clinically recognizable, the real challenge is not naming the spasm, but performing a rigorous differential diagnosis with conditions that can mimic it or coexist with it, including hemifacial spasm, central lesions at the cerebellopontine angle, peripheral nerve trauma, demyelinating disorders, and infiltrative or compressive neoplasms.
Starting from the broader category of peripherally induced movement disorders, the chapter emphasizes that abnormal involuntary movements can be triggered by trauma to cranial or peripheral nerves or roots, but may also be generated or modulated by central mechanisms. This principle obliges the clinician to keep the diagnostic horizon wide. Tumors of the cerebellopontine angle—such as vestibular schwannomas, meningiomas, or epidermoid tumors—have been reported as rare causes of hemifacial spasm, with mechanisms including vascular involvement, direct nerve compression, or brainstem distortion. The chapter extends this reasoning to the trigeminal system, highlighting that trigeminal schwannomas are uncommon but clinically relevant: they may present atypically, even as painless malocclusion with unilateral masticatory weakness, forcing dentistry and neurophysiology to intersect in the same diagnostic space. Neuroimaging remains essential, but must be interpreted inside a neurofunctional framework rather than as a stand-alone “explanation.”
A second major branch of differential diagnosis concerns demyelinating disease. Multiple sclerosis is reviewed as an archetype of multifocal CNS damage with variable clinical expression and imperfect diagnostic certainty. Importantly, trigeminal reflex testing—especially the jaw jerk and masseteric silent period—can reveal brainstem involvement in selected cases, sometimes when other reflexes appear normal. This creates a clinically meaningful dilemma: when trigeminal reflex abnormalities are detected, the clinician must determine whether they reflect central demyelination, peripheral motor branch dysfunction, or an alternative process.
The chapter then examines neoplastic and inflammatory disorders capable of producing facial spasms or perineural effects, including salivary gland tumors such as pleomorphic adenoma, whose imaging assessment (CT vs MRI) differs in soft-tissue delineation and evaluation of possible perineural invasion. Finally, the chapter introduces systemic and localized scleroderma, focusing on Morphea as the patient’s documented condition and discussing reports linking localized scleroderma/facial hemiatrophy to hemimasticatory spasm. On trigeminal clinical and electrophysiological grounds, focal demyelination of trigeminal motor branches and abnormal excitatory phenomena are proposed as plausible mechanisms.
The diagnostic “encrypted code” that organizes this complexity is identified as ephaptic transmission: pathological electrical communication between adjacent neural elements in the absence of normal synaptic contact. The chapter closes by positioning ephaptic mechanisms as a conceptual bridge between clinical spasm, reflex asymmetries, and neurophysiological interpretation, preparing the reader for the dedicated chapter on the two forms of electrical transmission between neurons.
-
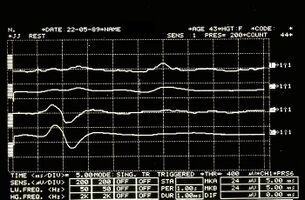
Figure 3: Mandibular reflex detected electrophysiologically on the right (upper traces) and left (lower traces) masseters.
-
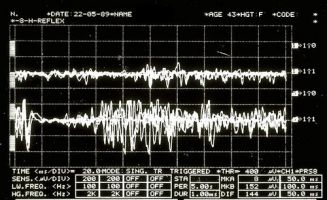
Figure 4: Mechanical silent period detected electrophysiologically on the right (upper overlapping traces) and left (lower overlapping traces) masseters
🧠 Three guiding questions (with essential answers)
1️⃣ Why is Hemimasticatory Spasm diagnostically challenging despite its recognizable clinical appearance? — Because the unilateral masticatory spasm can mimic or overlap with hemifacial spasm, demyelinating disorders, peripheral nerve injuries, or central lesions, making differential diagnosis essential to avoid incorrect etiological attribution and inappropriate treatment.
2️⃣ Why are trigeminal reflexes crucial in the differential diagnosis of masticatory movement disorders? — Because electrophysiological tests such as the jaw jerk and masseteric silent period can reveal functional asymmetries and conduction abnormalities that help distinguish central demyelination, peripheral motor branch involvement, and abnormal excitatory phenomena within the trigeminal system.
3️⃣ What role does ephaptic transmission play in the pathophysiology of Hemimasticatory Spasm? — Ephaptic transmission provides a mechanistic explanation for abnormal involuntary muscle activity by allowing pathological electrical coupling between adjacent neural elements, generating excitatory signals without normal synaptic mediation.
If you already have an approved account, click on your profile icon and return to the Book Index to read the complete chapter.
To access the full content, you must sign in via LinkedIn and request approval.
Bibliography & references
- Jankovic J. Peripherally induced movement disorders. Neurol Clin. 2009 Aug;27(3):821–832. DOI: 10.1016/j.ncl.2009.04.005.
- Lee SH, Rhee BA, Choi SK, Koh JS, Lim YJ. Cerebellopontine angle tumors causing hemifacial spasm: types, incidence, and mechanism in nine reported cases and literature review. Acta Neurochir (Wien). 2010 Nov;152(11):1901–1908. DOI: 10.1007/s00701-010-0796-1.
- Peker S, Ozduman K, Kiliç T, Pamir MN. Relief of hemifacial spasm after radiosurgery for intracanalicular vestibular schwannoma. Minim Invasive Neurosurg. 2004 Aug;47(4):235–237. DOI: 10.1055/s-2004-818485.
- Bertot BE, Lo Presti M, Stormes K, Raskin JS, Jea A, Chelius D, Lam S. Trigeminal schwannoma presenting with malocclusion: A case report and review of the literature. Surg Neurol Int. 2020 Aug 8;11:230. DOI: 10.25259/SNI_482_2019.
- Agarwal A. Intracranial trigeminal schwannoma. Neuroradiol J. 2015 Feb;28(1):36–41. DOI: 10.15274/NRJ-2014-10117.
- Kamińska J, Koper OM, Piechal K, Kemona H. Multiple sclerosis – etiology and diagnostic potential. Postepy Hig Med Dosw. 2017 Jun 30;71:551–563. DOI: 10.5604/01.3001.0010.3836.
- Yates SK, Brown WF. The human jaw jerk: electrophysiologic methods to measure the latency, normal values, and changes in multiple sclerosis. Neurology. 1981 May;31(5):632–634. DOI: 10.1212/WNL.31.5.632.
- Koyuncu M, Seşen T, Akan H, Ismailoglu AA, Tanyeri Y, Tekat A, Unal R, Incesu L. Comparison of computed tomography and magnetic resonance imaging in the diagnosis of parotid tumors. Otolaryngol Head Neck Surg. 2003 Dec;129(6):726–732. DOI: 10.1016/j.otohns.2003.07.009.
- Machado RA, Moubayed SPM, Khorsandi A, Hernandez-Prera JC, Urken ML. Intermittent facial spasms as the presenting sign of a recurrent pleomorphic adenoma. World J Clin Oncol. 2017 Feb 10;8(1):86–90. DOI: 10.5306/wjco.v8.i1.86.
- Amaral TN, Peres FA, Lapa AT, Marques-Neto JF, Appenzeller S. Neurologic involvement in scleroderma: a systematic review. Semin Arthritis Rheum. 2013 Dec;43(3):335–347. DOI: 10.1016/j.semarthrit.2013.05.002.
- James W, Berger T, Elston D. Andrews’ Diseases of the Skin: Clinical Dermatology. 10th ed. Saunders; 2005. p.171. ISBN: 0-7216-2921-0.
- Kim HJ, Jeon BS, Lee KW. Hemimasticatory spasm associated with localized scleroderma and facial hemiatrophy. Arch Neurol. 2000 Apr;57(4):576–580. DOI: 10.1001/archneur.57.4.576.