Levels of clinical observation: change of informational scale: differenze tra le versioni
Creata pagina con "{{main menu it |link to German= Hauptseite |link to Spanish= Página Principal |link to French= Page d'accueil |link to Italian= Indice Ψ |link to English= Index Ψ | no title = 1 }} <div class="chapter-content"> <div class="chapter-container"> = Levels of clinical observation: change of informational scale = <br> __TOC__ <html> <style> →TOC without frame: .toc { border: none !important; background: transparent !important; padding: 0 !important; margin:..." |
Nessun oggetto della modifica |
||
| Riga 1: | Riga 1: | ||
{{main menu | {{main menu en | ||
|link to German= Hauptseite | |link to German= Hauptseite | ||
|link to Spanish= Página Principal | |link to Spanish= Página Principal | ||
Versione delle 11:09, 24 gen 2026
Levels of clinical observation: change of informational scale
Abstract
Contemporary clinical practice is characterized by an increasing availability of heterogeneous data, collected at different levels of observation. However, diagnostic difficulty does not arise from the quantity of information, but from the failure to explicitly define the scales to which such information belongs and the way in which it is integrated.
This chapter introduces the concept of information scale as a level of observational resolution, distinct from the notion of clinical relevance, showing how the same phenomenon may appear stable or unstable depending on the scale at which it is analyzed. The change of scale is not treated as a neutral operation, but as an active act that redistributes information between functional contour, local detail, and system noise.
Through a clinical example centered on observation of the masticatory system, the chapter illustrates the progressive descent from clinical macroscopic observation to neuronal microscopy, passing through instrumental and mesoscopic levels. Along this path it emerges that increasing observational resolution inevitably entails a loss of global context and an increase in variability, which cannot be automatically interpreted as error or pathology.
The distinction between macroscopic, mesoscopic, and microscopic clinical observation allows trigeminal reflexes and neuromotor dynamics to be placed on an intermediate scale, in which the system is observed as a network of functional relations rather than as a set of isolated variables or as simple global behavior.
The chapter therefore proposes a view of diagnosis as a process dependent on the scale of observation, laying the foundations for clinical models capable of integrating variability, context, and system relations, and preparing the conceptual ground for discussing the limits of traditional classificatory criteria and for introducing mesoscopic and adimensional diagnostic quantities.
1. Why speak of scale in clinical practice
In the clinical domain, the problem is almost never the lack of data, but the heterogeneity of the levels to which those data belong. Patient-reported symptoms, clinical signs, instrumental parameters, neurophysiological measurements, and interpretative models do not exist on the same informational plane.
The clinician is constantly forced to move between different scales, often without this transition being made explicit. The result is an improper superimposition of information:
- fine-grained data interpreted with coarse categories
- complex phenomena compressed into simplifying labels
- precise measurements integrated into approximate conceptual models[1]
Speaking of levels of observation therefore means making explicit a practice that already exists, but has so far remained implicit and poorly formalized.[2]
---
2. What is meant by “information scale”
By scale one does not mean a hierarchy of importance, but a level of resolution with which a phenomenon is observed, described, and interpreted.[3]
Each scale is characterized by:
- type of accessible information
- observational instruments
- descriptive language
- degree of tolerable variability
- dominant type of error
A crucial point is that the same phenomenon may appear stable at one scale and unstable at another, without implying a contradiction.[4][5]
---
3. The main levels of observation in clinical practice
Without claiming to be exhaustive, some recurrent levels can be identified in clinical practice.
3.1 Phenomenological level (clinical-narrative)
This is the level of:
- reported symptom
- patient narrative
- temporal and subjective context
Here information is narrative, situated, and strongly context-dependent. Variability is high and cannot be reduced without loss of meaning.
This level is not less scientific: it is simply low in formal resolution, but high in semantic density.
---
3.2 Clinical-functional level
It includes:
- physical examination
- clinical tests
- functional assessments
The phenomenon is anchored to reproducible patterns, but remains dependent on the observer and execution conditions.
This is an intermediate scale: it reduces narrative complexity without yet entering fine instrumental measurement.[6]
---
3.3 Instrumental level
This is the domain of:
- measurements
- signals
- quantitative parameters
Information is high-resolution but localized. Here a classic problem emerges: precision of data does not guarantee precision of interpretation.
A signal is always correct within its own measurement space, but may become misleading if transferred without mediation to another scale.
---
3.4 Interpretative–modeling level
This is the level where:
- data are integrated
- relations are hypothesized
- clinical models take shape
This level does not directly observe the phenomenon, but reconstructs it. It is here that conflicts of meaning emerge when:
- models designed for one scale are applied to another
- internal coherence of the model is confused with clinical validity[2]
---
4. Change of scale as an active operation
The transition between scales is not automatic. It is an active operation that always entails:
- loss of information
- gain of structure
- introduction of assumptions
Each change of scale selects what remains visible and what is sacrificed.
This explains why:
- two clinicians may observe the same patient yet describe different phenomena
- an instrumental result may appear incoherent with the clinical picture
- stability emerges only at certain scales
It is important to emphasize that the relationship between observational scale and noise is neither linear nor univocal. Variability observed in a complex system does not depend exclusively on the intrinsic properties of the system itself, but also on the level of resolution at which it is observed.
At the macroscopic scale, reduced observational resolution integrates micro-variability into the global behavior of the system, making individual fluctuations difficult to distinguish. Consequently, observable noise appears reduced, despite the presence of underlying variability that continues to exist.
Descending toward finer observational scales, increased resolution makes visible system components that were previously integrated, revealing variability that cannot be automatically interpreted as error, but as a property dependent on the scale of observation.
Clinical example: descending scale in observation of the masticatory system
A particularly instructive example of information scale change is provided by clinical observation of the masticatory system, analyzed at progressively finer levels of resolution.
Clinical observation of occlusal–masticatory state
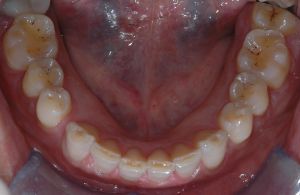
The clinician evaluates functional coordination, apparent efficiency of the gesture, and the presence of adaptations and compensations.
At this scale the system often appears stable and coherent, even in the presence of underlying alterations. High integration of information makes observation robust and poorly sensitive to local fluctuations. In this case, objective analysis of occlusal status is low-resolution but offers a broad view of the functional contour. In Figure 1, dental wear due to bruxism is evident and classification of the subject as a bruxer appears straightforward.
Surface EMG: interference pattern
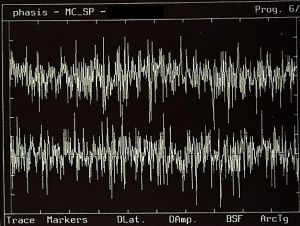
At this level, variations emerge that were not clinically evident at the higher scale. Greater resolution makes the signal more informative, but also more sensitive to noise[8] and measurement conditions.
Essentially, resolution increases but intrinsic system noise also increases. In the previously shown clinical case, EMG interference patterns provide indications about system state (discharge frequency, amplitudes, etc.) but begin to incorporate destabilizing noise, both technical and neurophysiological. We are in an intermediate phase between contour visualization, system noise, and data interpretation.[9]The result is diagnostic uncertainty.[10]
Neuromotor processes: recovery cycle of the mandibular reflex
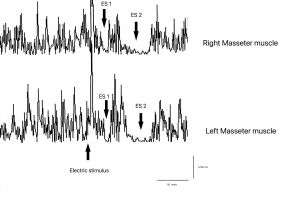
Unlike a single reflex response, the recovery cycle allows analysis of the temporal dynamics of the neuromotor system,[12] evaluating:
- excitability modulation
- recovery times
- inhibitory and facilitatory interactions
- central control of the reflex
At this scale information is no longer static, but intrinsically dynamic. Response variability does not represent an anomaly,[13] but a structural characteristic of neuromotor functioning.
Global functional context of the masticatory gesture is largely lost, while sensitivity to temporal fluctuations and system state conditions increases. In short, resolution increases as scale descends, but noise increases concurrently. The recovery cycle of the masseter inhibitory reflex provides major interpretative contribution to system state, but is very difficult to clean from noise[14] because variability of the evoking stimulus (electrical) and the masticatory force developed during maximal contraction of masticatory muscles do not allow extraction of a pure datum. Figure 3It is essential to clarify that experimental isolation of reflex circuits, as proposed in neurophysiological literature, does not aim to eliminate variability in an absolute sense, but to reduce noise components related to global motor behavior and higher functional context.
The descent in scale performed in the study of trigeminal reflexes allows control of macroscopic sources of variability, but inevitably makes intrinsic fluctuations of the central nervous system more evident, linked to excitability state, synaptic dynamics, and central modulation mechanisms.
Therefore, greater variability observed at this scale does not represent a methodological contradiction, but a direct consequence of increased observational resolution. What at higher scales was integrated into global system behavior emerges, at the mesoscopic level, as structural variability of neuromotor control.
Neuronal microdynamics: patch-clamp of mesencephalic nuclei
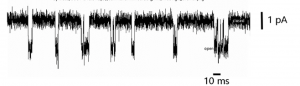
Ionic currents, neuronal firing, and spontaneous fluctuations of cellular activity are analyzed.
At this level variability cannot be interpreted as experimental error:[16] it constitutes an integral part of system functioning. The notion of stability, as understood at higher scales, loses meaning.[17]
At this observational scale the signal does not appear as a stable quantity, but as a succession of discrete and temporally fluctuating events. Amplitude variations do not represent instrumental noise, but stochastic opening and closing of individual ion channels.
In this example recorded currents have an amplitude of approximately 2 pA (2 × 10⁻¹² Ampere), corresponding to the passage of a few thousand elementary charges per millisecond. The extraordinary sensitivity of the method makes visible variability that, at higher scales, is completely integrated and therefore invisible.
It is important to note that the greater “instability” observed at this scale does not imply a real increase in system noise, but reflects loss of the global functional contour and direct emergence of biological microdynamics.
Considerations on scale change
This example shows how, descending progressively in scale:
- local observational resolution increases
- previously invisible fluctuations emerge
- global functional contour is progressively lost
Conversely, ascending in scale:
- the system regains coherence and apparent stability
- micro-variability is integrated or absorbed
- local noise loses clinical weight
Scale change does not eliminate information, but reorganizes it. Many interpretative discrepancies in clinical practice derive from attempts to apply stability criteria proper to one scale to observations made at another.
4.1 Clinical macroscopic, mesoscopic, and microscopic domains
In describing levels of clinical observation it is therefore useful to distinguish, in a purely conceptual way, three major scale domains.
Clinical macroscopic observation concerns global functional observation of the system: occlusal arrangement, masticatory gesture, adaptations, and compensations emerge at this scale as integrated and relatively stable properties.
Microscopic observation instead focuses on elementary mechanisms of the biological system, down to the level of the single cell or single ion channel. At this scale variability is high and constitutes an intrinsic property of functioning, making extraction of clinically stable quantities difficult.
Between these two extremes lies an intermediate scale, definable as mesoscopic, in which the system is observed neither as a global whole nor as a sum of elementary components, but as a dynamic network of functional relations.
Trigeminal reflexes and neuromotor recovery cycles represent typical examples of mesoscopic observations: they do not describe macroscopic behavior of the gesture, nor neuronal microdynamics, but the way the system organizes and modulates its activity over time.
At this scale, more than absolute magnitudes of individual variables, the relationships between quantities, their relative stability, and coherence of system behavior become relevant.
- ↑ Norman G, Eva K. Diagnostic error and clinical reasoning. Medical Education, 2010
- ↑ 2,0 2,1 Bogen J, Woodward J. Saving the phenomena. Philosophical Review, 1988
- ↑ Marr D. ''Vision: A Computational Investigation into the Human Representation and Processing of Visual Information''. MIT Press; 1982.
- ↑ Goldenfeld N, Kadanoff LP. Simple lessons from complexity. ''Science''. 1999;284(5411):87–89.
- ↑ Kitano H. Systems biology: a brief overview. ''Science''. 2002;295(5560):1662–1664.
- ↑ Sturmberg JP, Martin CM. Complexity and health: moving from linear to complex thinking. Journal of Evaluation in Clinical Practice, 2009
- ↑ De Luca CJ. The use of surface electromyography in biomechanics. Journal of Applied Biomechanics, 1997.
- ↑ Farina D, Merletti R, Enoka RM. The extraction of neural strategies from the surface EMG. Journal of Applied Physiology, 2004.
- ↑ Enoka RM, Duchateau J. Muscle fatigue: what, why and how it influences muscle function. Journal of Physiology, 2008.
- ↑ Türker KS. Electrophysiological methods in orofacial pain research. ''Journal of Oral Rehabilitation''. 2002;29:918–930.
- ↑ Cruccu G, Inghilleri M, Berardelli A, Manfredi M. Pathophysiology of the masseter inhibitory reflex. ''Electroencephalography and Clinical Neurophysiology''. 1984.
- ↑ Türker KS, Jenkins M. Reflex responses of human jaw muscles to mechanical stimulation. ''Experimental Brain Research''. 2000.
- ↑ Türker KS. Reflex control of human jaw muscles. ''Critical Reviews in Oral Biology & Medicine''. 2002.
- ↑ Svensson P, Graven-Nielsen T. Craniofacial muscle pain: review of mechanisms and clinical implications. ''Journal of Orofacial Pain''. 2001.
- ↑ Neher E, Sakmann B. Single-channel currents recorded from membrane of denervated frog muscle fibres. ''Nature''. 1976.
- ↑ Faisal AA, Selen LPJ, Wolpert DM. Noise in the nervous system. ''Nature Reviews Neuroscience''. 2008.
- ↑ Marr D. ''Vision: A Computational Investigation into the Human Representation and Processing of Visual Information''. MIT Press; 1982.